6-K
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 6-K
REPORT OF FOREIGN PRIVATE ISSUER
PURSUANT TO RULE 13A-16 OR 15D-16 UNDER
THE SECURITIES EXCHANGE ACT OF 1934
For the month of January 2026
Commission File Number: 001-40010
Pharvaris N.V.
(Translation of registrant’s name into English)
Emmy Noetherweg 2
2333 BK Leiden
The Netherlands
(Address of principal executive office)
Indicate by check mark whether the registrant files or will file annual reports under cover of Form 20-F or Form 40-F.
Form 20-F ☒ Form 40-F ☐
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(1): ☐
Note: Regulation S-T Rule 101(b)(1) only permits the submission in paper of a Form 6-K if submitted solely to provide an attached annual report to security holders.
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(7): ☐
Note: Regulation S-T Rule 101(b)(7) only permits the submission in paper of a Form 6-K if submitted to furnish a report or other document that the registrant foreign private issuer must furnish and make public under the laws of the jurisdiction in which the registrant is incorporated, domiciled or legally organized (the registrant’s “home country”), or under the rules of the home country exchange on which the registrant’s securities are traded, as long as the report or other document is not a press release, is not required to be and has not been distributed to the registrant’s security holders, and, if discussing a material event, has already been the subject of a Form 6-K submission or other Commission filing on EDGAR.
PHARVARIS N.V.
On January 12, 2026, Pharvaris N.V. (the “Company”) issued a press release. The press release is attached as Exhibit 99.1 hereto and is incorporated by reference herein. Also on January 12, 2026, the Company made available an investor presentation on its website. A copy of the investor presentation is attached hereto as Exhibit 99.2.
Exhibit 99.1 to this Report on Form 6-K shall be deemed to be incorporated by reference into the registration statements on Form F-3 (Registration Number 333-273757, 333-277705 and 333-278650) and Form S-8 (Registration Number 333-252897). Exhibit 99.2 to this Report on Form 6-K shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”) or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference in any filing under the Securities Act of 1933, as amended or the Exchange Act.
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
|
|
|
|
PHARVARIS N.V. |
|
|
Date: January 12, 2026 |
By: |
/s/ Berndt Modig |
|
Name: |
Berndt Modig |
|
Title: |
Chief Executive Officer |
EXHIBIT INDEX
EX-99.1

Exhibit 99.1
Pharvaris Outlines 2026 Strategic Priorities
•Topline data from CHAPTER-3, a pivotal study of deucrictibant for prophylactic treatment of HAE attacks, anticipated in 3Q2026
•Preparation of NDA dossier of deucrictibant for on-demand treatment of HAE attacks ongoing; timeline remains on-track for filing in 1H2026
•Recruitment ongoing in CREAATE, a pivotal study of deucrictibant for the prophylactic and on-demand treatment of AAE-C1INH attacks
•Estimated cash runway into 1H2027
ZUG, Switzerland, January 12, 2026 – Pharvaris (Nasdaq: PHVS), a late-stage biopharmaceutical company developing novel, oral bradykinin B2 receptor antagonists to help address unmet needs of those living with bradykinin-mediated diseases such as hereditary angioedema (HAE) and acquired angioedema due to C1 inhibitor deficiency (AAE-C1INH), today outlined its strategic priorities for 2026.
“The readout of Pharvaris’ first pivotal Phase 3 study, RAPIDe-3, in December was the culmination of a decade of scientific rigor, operational and financial diligence, executional excellence, and, most importantly, community engagement and commitment,” said Berndt Modig, Chief Executive Officer of Pharvaris. “The data reported in December build upon Pharvaris’ legacy in HAE drug development, and we believe demonstrate deucrictibant’s potentially differentiated profile and potential to become a new standard of care for on-demand HAE treatment of attacks. Our team’s ability to conduct the most diverse Phase 3 on-demand study in HAE, by including previously underserved regions and subgroups, and improve upon the outcomes of the RAPIDe-1 Phase 2 study further bolsters our confidence in the clinical execution of the CHAPTER-3 prophylactic study, for which the timing of anticipated data readout has now been refined to the third quarter of 2026.”
2026 Strategic Priorities
Long-term Prophylaxis of HAE Attacks
•Topline data from CHAPTER-3 (NCT06669754) anticipated 3Q2026. CHAPTER-3 is a randomized, double-blind, placebo-controlled Phase 3 study of orally administered deucrictibant extended-release tablet for the prophylaxis against angioedema attacks in adults and adolescents (12 years and older) with HAE. The study aims to enroll approximately 81 participants randomized in a 2:1 ratio to receive deucrictibant extended-release tablet (40 mg), which is the
intended commercial dosage, or placebo, once daily for 24 weeks. Pharvaris anticipates announcing topline data of CHAPTER-3 in the third quarter of 2026.
•Enrollment in CHAPTER-4 (NCT06679881) progressing as planned. CHAPTER-4 is a long-term, open-label extension study of orally administered deucrictibant extended-release tablet (40 mg/day) for the prophylactic treatment of HAE attacks. The goal of the study is to evaluate the long-term safety and effectiveness of deucrictibant extended-release tablet in the prophylactic treatment of HAE attacks.
•Completed CHAPTER-1 (NCT05047185); final data recently presented. Final Results from the randomized portion and the long-term open-label portion of the study demonstrated that deucrictibant was well tolerated for up to approximately three years. The mean rate of HAE attacks was reduced by deucrictibant within the first week of treatment and remained low for up to approximately 34 months, with an overall mean monthly on-treatment attack rate of 0.12 throughout the completed open-label extension portion of the study.
On-demand Treatment of HAE Attacks
•RAPIDe-3 (NCT06343779) met primary endpoint and all secondary efficacy endpoints with statistical significance. Outcomes from RAPIDe-3, a pivotal global Phase 3 study evaluating orally administered deucrictibant immediate-release capsule (20 mg) for the on-demand treatment of HAE attacks in adults and adolescents (12 years and older), confirming the potential of deucrictibant’s differentiated profile for the on-demand treatment of HAE attacks. The primary endpoint, median time to onset of symptom relief, was achieved in 1.28 hours, significantly faster versus placebo (p<0.0001), and deucrictibant was well tolerated. Pharvaris plans to present additional efficacy, safety, and patient experience data at upcoming medical congresses.
•Filing of U.S. New Drug Application (NDA) of deucrictibant for the on-demand treatment of HAE attacks anticipated 1H2026. Pharvaris is preparing the dossier for deucrictibant’s NDA filing. The data from RAPIDe-3 and RAPIDe-2 will serve as the basis for marketing authorization applications, which are planned to be filed starting in the first half of 2026.
Clinical Development of Deucrictibant in AAE-C1 INH
•CREAATE (NCT06669754) study progressing as planned. Pharvaris initiated CREAATE, a global, pivotal Phase 3 study of deucrictibant for the prophylactic and on-demand treatment of AAE-C1INH attacks, in November 2025. CREAATE assesses the efficacy and safety of deucrictibant in people living with AAE-C1INH. In part 1 of CREAATE, participants receive either deucrictibant extended-release tablet (40 mg) or placebo once daily for the prophylactic
treatment of AAE-C1INH attacks. In part 2 of CREAATE, participants treat two attacks in a cross-over fashion, one attack with deucrictibant immediate-release capsule (20 mg) and one with placebo according to a randomized treatment sequence, for the on-demand treatment of AAE-C1INH attacks. Part 3 of CREAATE is the open-label extension portion of the study assessing the long-term safety and effectiveness of deucrictibant immediate-release capsule (20 mg) for on-demand treatment.
Business Updates
Corporate
•Cash runway into 1H2027. Pharvaris remains diligent in its operational management and is focusing on late-stage clinical development programs and commercial preparedness for the potential launch of deucrictibant.
•Pharvaris recently added to Nasdaq Biotechnology Index (NBI). In December 2025, Pharvaris was added to the NBI. Companies in the NBI must meet eligibility requirements, including minimum market capitalization, average daily trading volume and seasoning as a public company, among other criteria. The NBI is evaluated annually in December and is calculated under a modified capitalization-weighted methodology.
Upcoming Participation at Investor Conferences
•Oppenheimer 36th Annual Healthcare Life Sciences Conference. Virtual, February 25-26, 2026.
•Format: Fireside Chat
Date, time: Thursday, February 26, 2026, 9:20-9:50 a.m. EST
•The Citizens Life Sciences Conference. Miami, FL, March 10-11, 2026.
•Format: Fireside Chat
Date, time: Tuesday, March 10, 2026, 11:20 a.m. EST
•Leerink Global Healthcare Conference 2026. Miami, FL, March 8-11, 2026.
•Format: Fireside Chat
Date, time: Wednesday, March 11, 2026, 9:20 a.m. EST
Live audio webcasts of the presentations will be available on the Investors section of the Pharvaris website at: https://ir.pharvaris.com/news-events/events-presentations. The audio replays will be available on Pharvaris’ website for 30 days following the presentation.
Upcoming Presentations at Medical Congresses
•Western Society of Allergy, Asthma & Immunology (WSAAI) 63rd Annual Scientific Session. Wailea, HI, February 1-5, 2026. Details of the accepted presentation at WSAAI are as follows:
•Title: Long-Term Safety and Efficacy of Oral Deucrictibant for Prophylaxis in Hereditary Angioedema: Results of the Phase 2 CHAPTER-1 Open-Label Extension Study
Presenter: Michael E. Manning, M.D.
Date, time: Wednesday, February 4, 2026, 7:00-8:00 a.m. HST (12:00-1:00 p.m. EST) and 11:30 a.m.-12:00 p.m. HST (4:30-5:00 p.m. EST)
•American Academy of Allergy, Asthma & Immunology (AAAAI) 2026 Annual Meeting. Philadelphia, PA, February 27-March 2, 2026. Details for the accepted presentations at AAAAI are as follows:
•Title: A Novel Kinin Biomarker Assay for Characterization of Different Types of Bradykinin-Mediated Angioedema
Presenter: Evangelia Pardali, Ph.D.
Poster Number: 078
Date, time: Friday, February 27, 2026, 2:45-3:45 p.m. EST
•Title: Content Validity of the Angioedema syMptom Rating scAle (AMRA) to Assess Symptoms of Hereditary Angioedema Attacks
Presenter: Teresa Caballero, M.D., Ph.D.
Poster Number: 154
Date, time: Friday, February 27, 2026, 2:45-3:45 p.m. EST
•Title: Long-Term Prophylactic Treatment with Oral Deucrictibant Improved Health-Related Quality of Life in Participants with Hereditary Angioedema: Final Results of the Phase 2 CHAPTER-1 Open-Label Extension Study
Presenter: Michael E. Manning, M.D.
Poster Number: 159
Date, time: Friday, February 27, 2026, 2:45-3:45 p.m. EST
•Title: Oral Deucrictibant Immediate-Release Capsule in Treatment of Hereditary Angioedema Attacks: Results of the Phase 3 RAPIDe-3 Study
Presenter: Marc A. Riedl, M.D., M.S.
Featured Poster Number: 831
Date, time: Sunday, March 1, 2026, 3:30-5:00 p.m. EST
•Title: Long-Term Safety and Efficacy of Oral Deucrictibant for Prophylaxis in Hereditary Angioedema: Final Results of the Phase 2 CHAPTER-1 Open-Label Extension Study
Presenter: John Anderson, M.D.
Featured Poster Number: 832
Date, time: Sunday, March 1, 2026, 3:30-5:00 p.m. EST
•Title: Sustained Therapeutic Exposure with Once-Daily Oral Deucrictibant Extended-Release Tablet for Prophylaxis of Hereditary Angioedema Attacks
Presenter: Zhi-Yi Zhang, Ph.D.
Featured Poster Number: 834
Date, time: Sunday, March 1, 2026, 3:30-5:00 p.m. EST
The posters will be available on the Investors section of the Pharvaris website at: https://ir.pharvaris.com/news-events/events-presentations.
About Deucrictibant
Deucrictibant is a novel, potent, orally bioavailable small-molecule bradykinin B2 receptor antagonist currently in clinical development. Deucrictibant is being investigated for its potential to prevent the occurrence of bradykinin-mediated angioedema attacks and to treat the manifestations of attacks if/when they occur by inhibiting bradykinin signaling through the bradykinin B2 receptor. Pharvaris is developing two formulations of deucrictibant for oral administration: an extended-release tablet to enable sustained absorption and efficacy as prophylactic treatment, and an immediate-release capsule to enable rapid onset of activity for on-demand treatment. Deucrictibant has been granted orphan drug designation for the treatment of bradykinin-mediated angioedema by the U.S. Food and Drug Administration, the European Commission, and Swissmedic.
About Pharvaris
Pharvaris is a late-stage biopharmaceutical company developing novel, oral bradykinin B2 receptor antagonists to help address unmet needs in bradykinin-mediated conditions, including all types of bradykinin-mediated angioedema. Pharvaris’ aspiration is to offer therapies with injectable-like efficacy™, a well-tolerated profile, and the convenience of oral administration to prevent and treat bradykinin-mediated angioedema attacks. By delivering on this aspiration, Pharvaris aims to provide a new standard of care in bradykinin-mediated angioedema. Pharvaris is preparing global marketing authorization applications for deucrictibant immediate-release capsule as an on-demand treatment of HAE attacks, and a global pivotal Phase 3 study of deucrictibant extended-release tablet for the prevention of
HAE attacks (CHAPTER-3) is ongoing with topline data anticipated in the third quarter of 2026. In addition, CREAATE is an ongoing Phase 3 study of deucrictibant for the prophylactic and on-demand treatment of AAE-C1INH attacks. For more information, visit https://pharvaris.com/.
Forward Looking Statements
This press release contains certain forward-looking statements that involve substantial risks and uncertainties. All statements contained in this press release that do not relate to matters of historical fact should be considered forward-looking statements, including, without limitation, statements relating to our future plans, studies and trials, and any statements containing the words “believe,” “anticipate,” “expect,” “estimate,” “may,” “could,” “should,” “would,” “will,” “intend” and similar expressions. These forward-looking statements are based on management’s current expectations, are neither promises nor guarantees, and involve known and unknown risks, uncertainties and other important factors that may cause Pharvaris’ actual results, performance or achievements to be materially different from its expectations expressed or implied by the forward-looking statements. Such risks include but are not limited to the following: uncertainty in the outcome of our interactions with regulatory authorities, including the FDA; the expected timing, progress, or success of our clinical development programs, especially for deucrictibant immediate-release capsules and deucrictibant extended-release tablets, which are in late-stage global clinical trials; our ability to replicate the efficacy and safety demonstrated in the RAPIDe-1, RAPIDe-2, RAPIDe-3, and CHAPTER-1 Phase 2 and Phase 3 studies in ongoing and future nonclinical studies and clinical trials, such as CHAPTER-3, and CREAATE; the timing and outcome of regulatory approvals, including the timing and outcome of our planned submission of an NDA with the FDA in the first half of 2026 for the on-demand treatment of acute attacks of HAE; risks arising from epidemic diseases, which may adversely impact our business, nonclinical studies, and clinical trials; our ability to potentially use deucrictibant for alternative purposes, for example to treat C1-INH deficiency (AAE-C1INH); the value of our ordinary shares; the timing, costs and other limitations involved in obtaining regulatory approval for our product candidates, or any other product candidate that we may develop in the future; our ability to establish commercial capabilities or enter into agreements with third parties to market, sell, and distribute our product candidates; our ability to compete in the pharmaceutical industry, including with respect to existing therapies, emerging potentially competitive therapies and with competitive generic products; our ability to market, commercialize and achieve market acceptance for our product candidates; our ability to produce sufficient amounts of drug product candidates for commercialization; our ability to raise capital when needed and on acceptable terms; regulatory developments in the United States, the European Union and other jurisdictions; our ability to protect our intellectual property and know-how and operate our business without infringing the intellectual property
rights or regulatory exclusivity of others; our ability to manage negative consequences from changes in applicable laws and regulations, including tax laws (including the Biosecure Act), our ability to maintain an effective system of internal control over financial reporting; changes and uncertainty in general market conditions; disruptions at the FDA and other agencies; changes and uncertainty in general market, political and economic conditions, including as a result of inflation and geopolitical conflicts; changes in regulations and customs, tariffs and trade barriers; and the other factors described under the headings “Cautionary Statement Regarding Forward-Looking Statements” and “Item 3. Key Information—D. Risk Factors” in our Annual Report on Form 20-F and other periodic filings with the U.S. Securities and Exchange Commission. These and other important factors could cause actual results to differ materially from those indicated by the forward-looking statements made in this press release. Any such forward-looking statements represent management’s estimates as of the date of this press release. New risks and uncertainties may emerge from time to time, and it is not possible to predict all risks and uncertainties. While Pharvaris may elect to update such forward-looking statements at some point in the future, Pharvaris disclaims any obligation to do so, even if subsequent events cause its views to change. These forward-looking statements should not be relied upon as representing Pharvaris’ views as of any date subsequent to the date of this press release.
Contact
Maggie Beller
Executive Director, Head of Corporate and Investor Communications
maggie.beller@pharvaris.com

Pioneering science for patient choice Corporate Presentation January 2026 Exhibit 99.2

Disclaimer This Presentation contains certain “forward‐looking statements” within the meaning of the federal securities laws that involve substantial risks and uncertainties. All statements contained in this Presentation that do not relate to matters of historical fact should be considered forward-looking statements, including, without limitation, statements relating to our future plans, studies and trials, and any statements containing the words “believe,” “anticipate,” “expect,” “estimate,” “may,” “could,” “should,” “would,” “will,” “intend” and similar expressions. We have based these forward-looking statements largely on our current expectations and projections about future events and trends that we believe may affect our financial condition, results of operations, business strategy and financial needs. Such forward-looking statements are neither promises nor guarantees, and involve known and unknown risks, uncertainties and other important factors which may cause our actual results, financial condition, performance or achievements, or industry results, to be materially different from any future results, performance or achievements expressed or implied by such forward-looking statements. Factors that might cause such a difference include, but are not limited to, uncertainty in the outcome of our interactions with regulatory authorities, including the FDA; the expected timing, progress, or success of our clinical development programs, especially for deucrictibant immediate-release capsules and deucrictibant extended-release tablets, which are in late-stage global clinical trials; our ability to replicate the efficacy and safety demonstrated in the RAPIDe-1, RAPIDe-2, RAPIDe-3, and CHAPTER-1 Phase 2 and Phase 3 studies in ongoing and future nonclinical studies and clinical trials, such as CHAPTER-3 and CREAATE; the timing and outcome of regulatory approvals, including the timing and outcome of our planned submission of an NDA with the FDA in the first half of 2026 for the on-demand treatment of acute attacks of HAE; risks arising from epidemic diseases, which may adversely impact our business, nonclinical studies, and clinical trials; our ability to potentially use deucrictibant for alternative purposes, for example to treat C1-INH deficiency (AAE-C1INH); the value of our ordinary shares; the timing, costs and other limitations involved in obtaining regulatory approval for our product candidates, or any other product candidate that we may develop in the future; our ability to establish commercial capabilities or enter into agreements with third parties to market, sell, and distribute our product candidates; our ability to compete in the pharmaceutical industry, including with respect to existing therapies, emerging potentially competitive therapies and with competitive generic products; our ability to market, commercialize and achieve market acceptance for our product candidates; our ability to produce sufficient amounts of drug product candidates for commercialization; our ability to raise capital when needed and on acceptable terms; regulatory developments in the United States, the European Union and other jurisdictions; our ability to protect our intellectual property and know-how and operate our business without infringing the intellectual property rights or regulatory exclusivity of others; our ability to manage negative consequences from changes in applicable laws and regulations, including tax laws (including the Biosecure Act); our ability to maintain an effective system of internal control over financial reporting; changes and uncertainty in general market conditions; disruptions at the FDA and other agencies; changes and uncertainty in general market, political and economic conditions, including as a result of inflation and geopolitical conflicts; changes in regulations and customs, tariffs and trade barriers; and the other factors described under the headings "Cautionary Statement Regarding Forward-Looking Statements" and "Item 3. Key Information--D. Risk Factors" in our Annual Report on Form 20-F and other periodic filings with the Securities and Exchange Commission. New risks and uncertainties may emerge from time to time, and it is not possible to predict all risks and uncertainties. We undertake no obligation to publicly update any forward-looking statements, whether as a result of new information, future events or otherwise, except as required by law. This presentation includes data for an investigational product not yet approved by regulatory authorities. Certain information contained in this Presentation relates to or is based on studies, publications, surveys and other data obtained from third-party sources and the Company’s own internal estimates and research. While the Company believes these third-party sources to be reliable as of the date of this presentation, it has not independently verified, and makes no representation as to the adequacy, fairness, accuracy or completeness of, any information obtained from third-party sources. In addition, all of the market data included in this Presentation involves a number of assumptions and limitations, and there can be no guarantee as to the accuracy or reliability of such assumptions. Finally, while we believe our own internal research is reliable, such research has not been verified by any independent source.
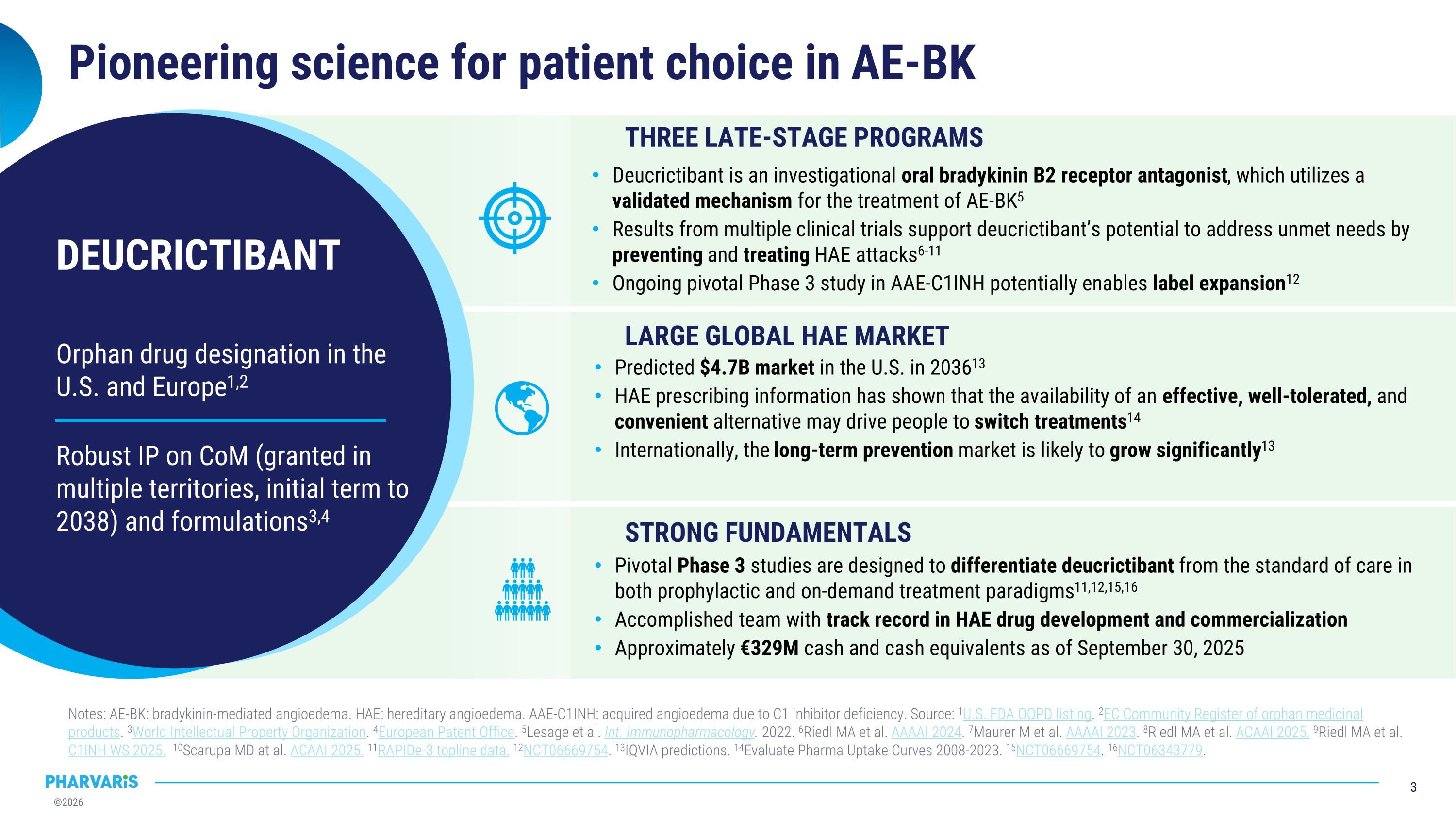
Orphan drug designation in the U.S. and Europe1,2 Robust IP on CoM (granted in multiple territories, initial term to 2038) and formulations3,4 DEUCRICTIBANT Pioneering science for patient choice in AE-BK Notes: AE-BK: bradykinin-mediated angioedema. HAE: hereditary angioedema. AAE-C1INH: acquired angioedema due to C1 inhibitor deficiency. Source: 1U.S. FDA OOPD listing. 2EC Community Register of orphan medicinal products. 3World Intellectual Property Organization. 4European Patent Office. 5Lesage et al. Int. Immunopharmacology. 2022. 6Riedl MA et al. AAAAI 2024. 7Maurer M et al. AAAAI 2023. 8Riedl MA et al. ACAAI 2025. 9Riedl MA et al. C1INH WS 2025. 10Scarupa MD at al. ACAAI 2025. 11RAPIDe-3 topline data. 12NCT06669754. 13IQVIA predictions. 14Evaluate Pharma Uptake Curves 2008-2023. 15NCT06669754. 16NCT06343779. THREE LATE-STAGE PROGRAMS LARGE GLOBAL HAE MARKET STRONG FUNDAMENTALS Deucrictibant is an investigational oral bradykinin B2 receptor antagonist, which utilizes a validated mechanism for the treatment of AE-BK5 Results from multiple clinical trials support deucrictibant’s potential to address unmet needs by preventing and treating HAE attacks6-11 Ongoing pivotal Phase 3 study in AAE-C1INH potentially enables label expansion12 Predicted $4.7B market in the U.S. in 203613 HAE prescribing information has shown that the availability of an effective, well-tolerated, and convenient alternative may drive people to switch treatments14 Internationally, the long-term prevention market is likely to grow significantly13 Pivotal Phase 3 studies are designed to differentiate deucrictibant from the standard of care in both prophylactic and on-demand treatment paradigms11,12,15,16 Accomplished team with track record in HAE drug development and commercialization Approximately €329M cash and cash equivalents as of September 30, 2025
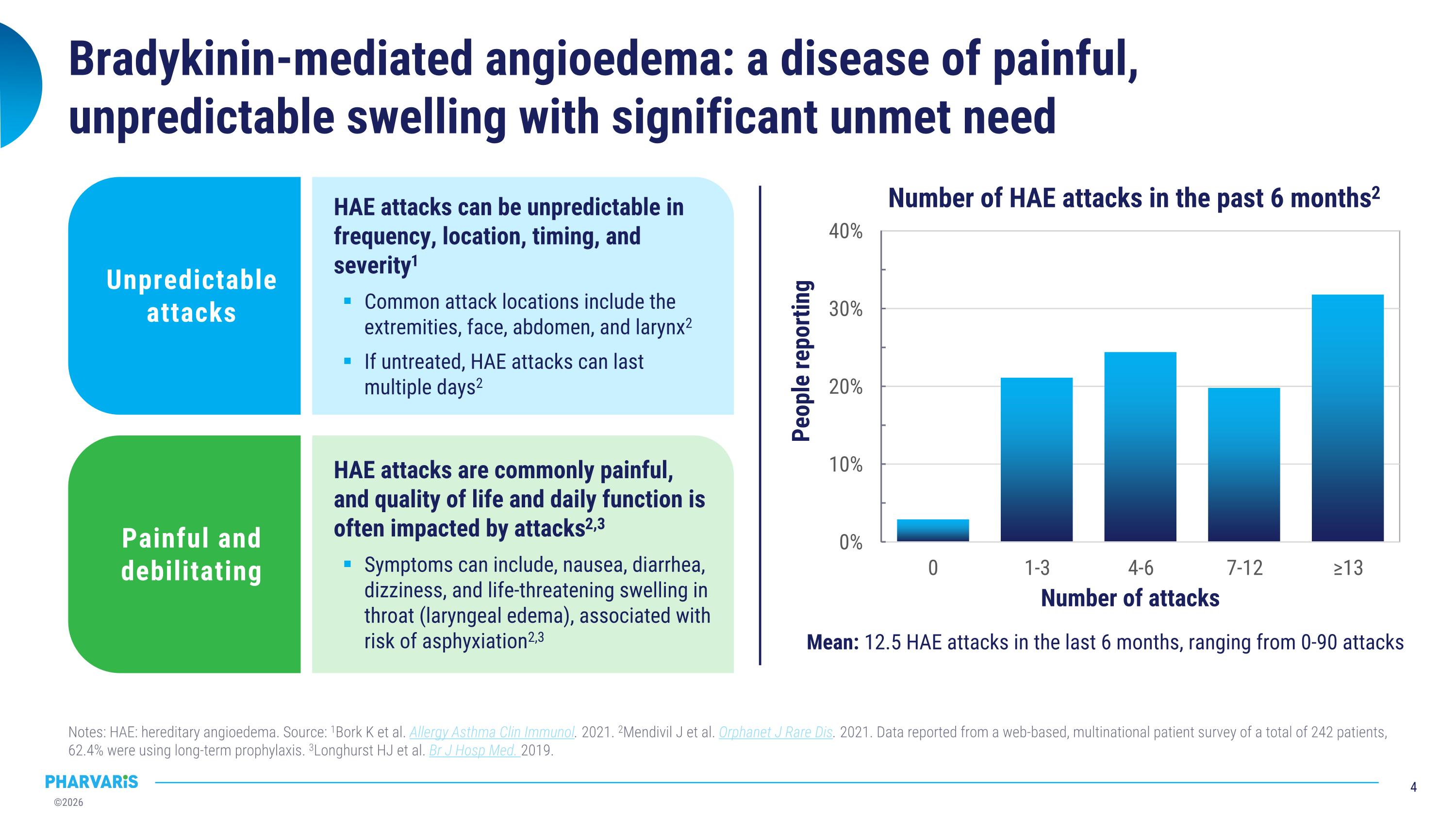
Bradykinin-mediated angioedema: a disease of painful, unpredictable swelling with significant unmet need Notes: HAE: hereditary angioedema. Source: 1Bork K et al. Allergy Asthma Clin Immunol. 2021. 2Mendivil J et al. Orphanet J Rare Dis. 2021. Data reported from a web-based, multinational patient survey of a total of 242 patients, 62.4% were using long-term prophylaxis. 3Longhurst HJ et al. Br J Hosp Med. 2019. Mean: 12.5 HAE attacks in the last 6 months, ranging from 0-90 attacks HAE attacks are commonly painful, and quality of life and daily function is often impacted by attacks2,3 Symptoms can include, nausea, diarrhea, dizziness, and life-threatening swelling in throat (laryngeal edema), associated with risk of asphyxiation2,3 Painful and debilitating HAE attacks can be unpredictable in frequency, location, timing, and severity1 Common attack locations include the extremities, face, abdomen, and larynx2 If untreated, HAE attacks can last multiple days2 Unpredictable attacks
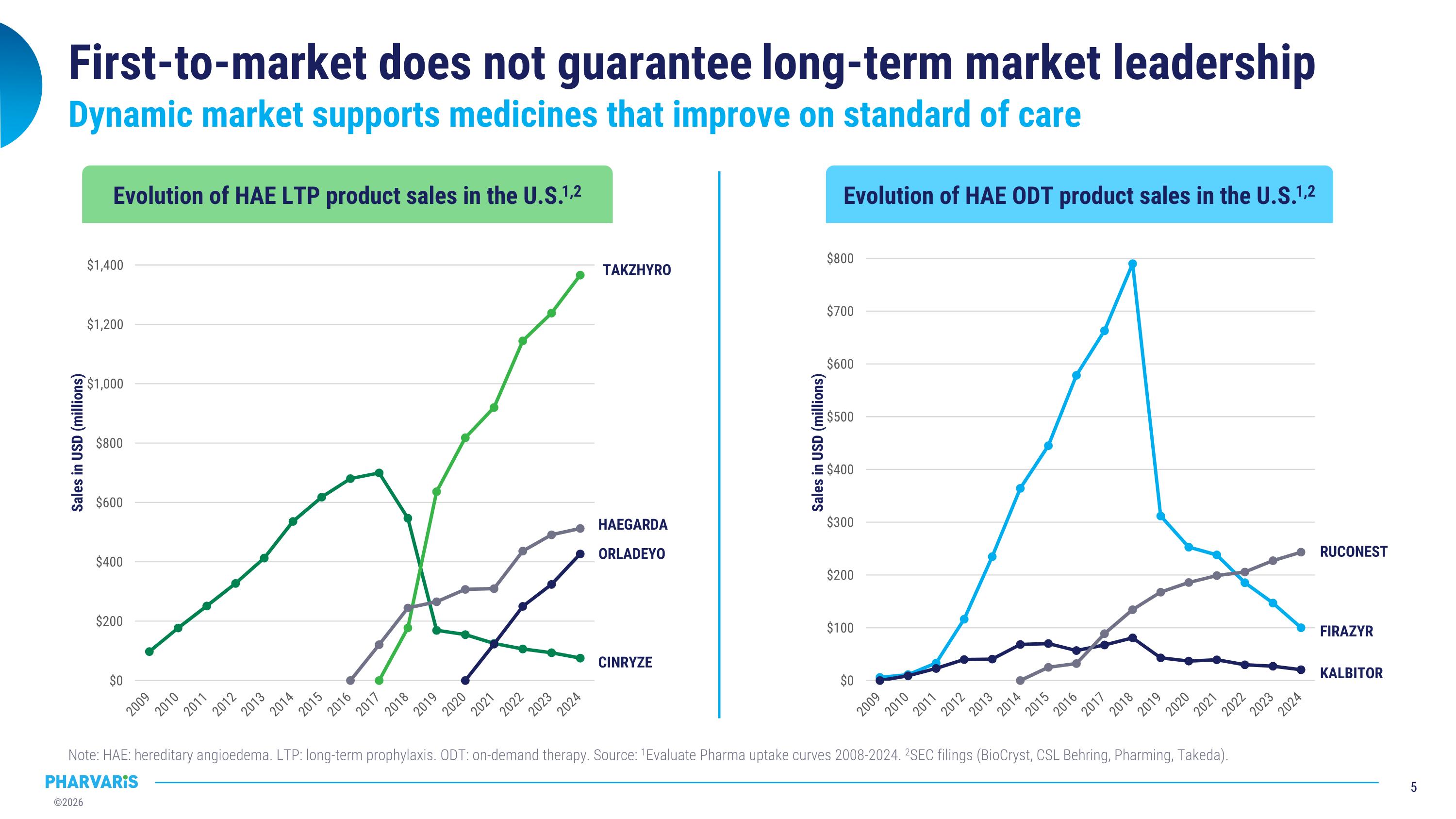
First-to-market does not guarantee long-term market leadership Note: HAE: hereditary angioedema. LTP: long-term prophylaxis. ODT: on-demand therapy. Source: 1Evaluate Pharma uptake curves 2008-2024. 2SEC filings (BioCryst, CSL Behring, Pharming, Takeda). Dynamic market supports medicines that improve on standard of care TAKZHYRO HAEGARDA ORLADEYO RUCONEST FIRAZYR CINRYZE KALBITOR Evolution of HAE LTP product sales in the U.S.1,2 Evolution of HAE ODT product sales in the U.S.1,2
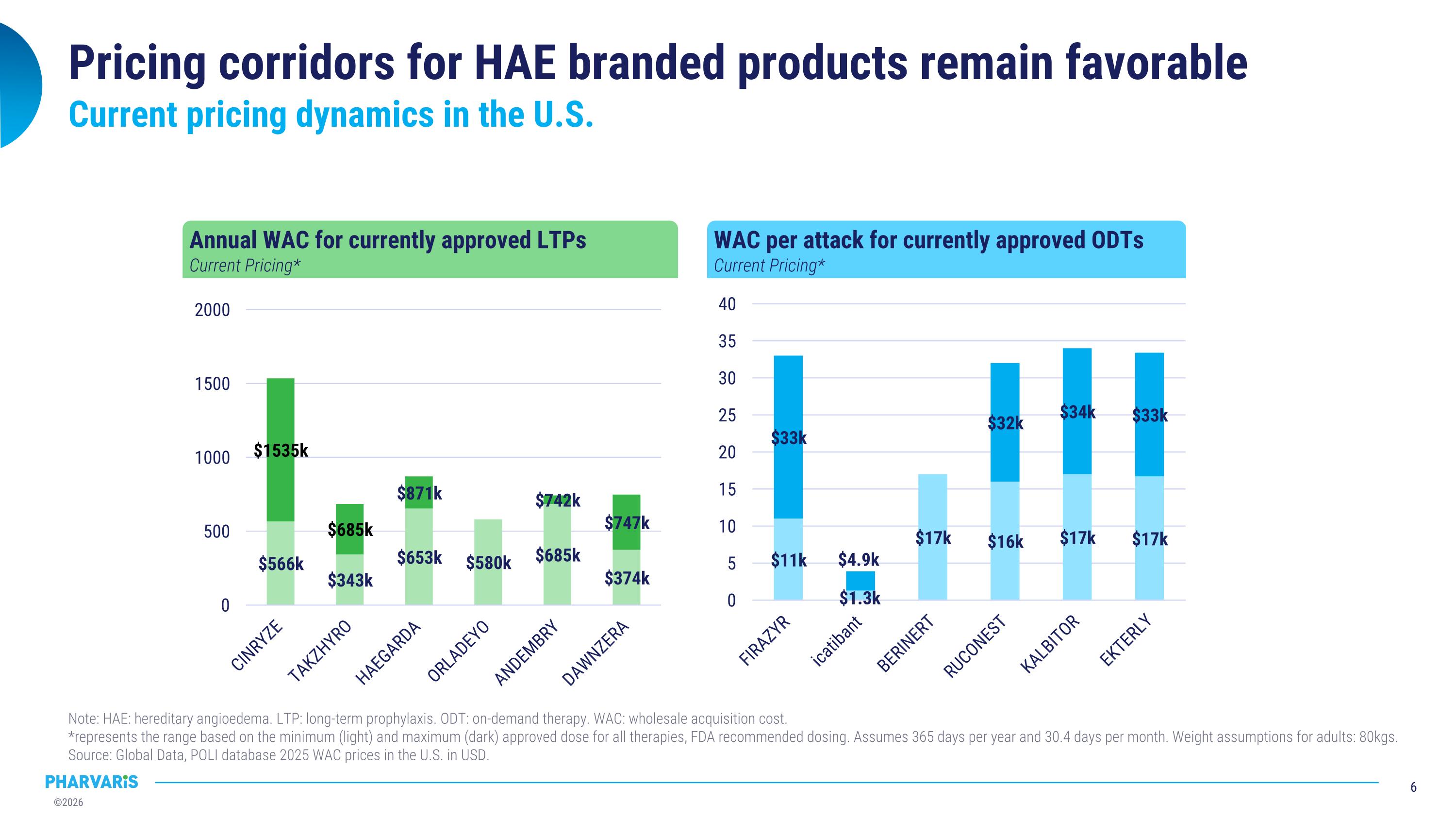
Pricing corridors for HAE branded products remain favorable Current pricing dynamics in the U.S. Annual WAC for currently approved LTPs Current Pricing* WAC per attack for currently approved ODTs Current Pricing* Note: HAE: hereditary angioedema. LTP: long-term prophylaxis. ODT: on-demand therapy. WAC: wholesale acquisition cost. �*represents the range based on the minimum (light) and maximum (dark) approved dose for all therapies, FDA recommended dosing. Assumes 365 days per year and 30.4 days per month. Weight assumptions for adults: 80kgs. �Source: Global Data, POLI database 2025 WAC prices in the U.S. in USD.
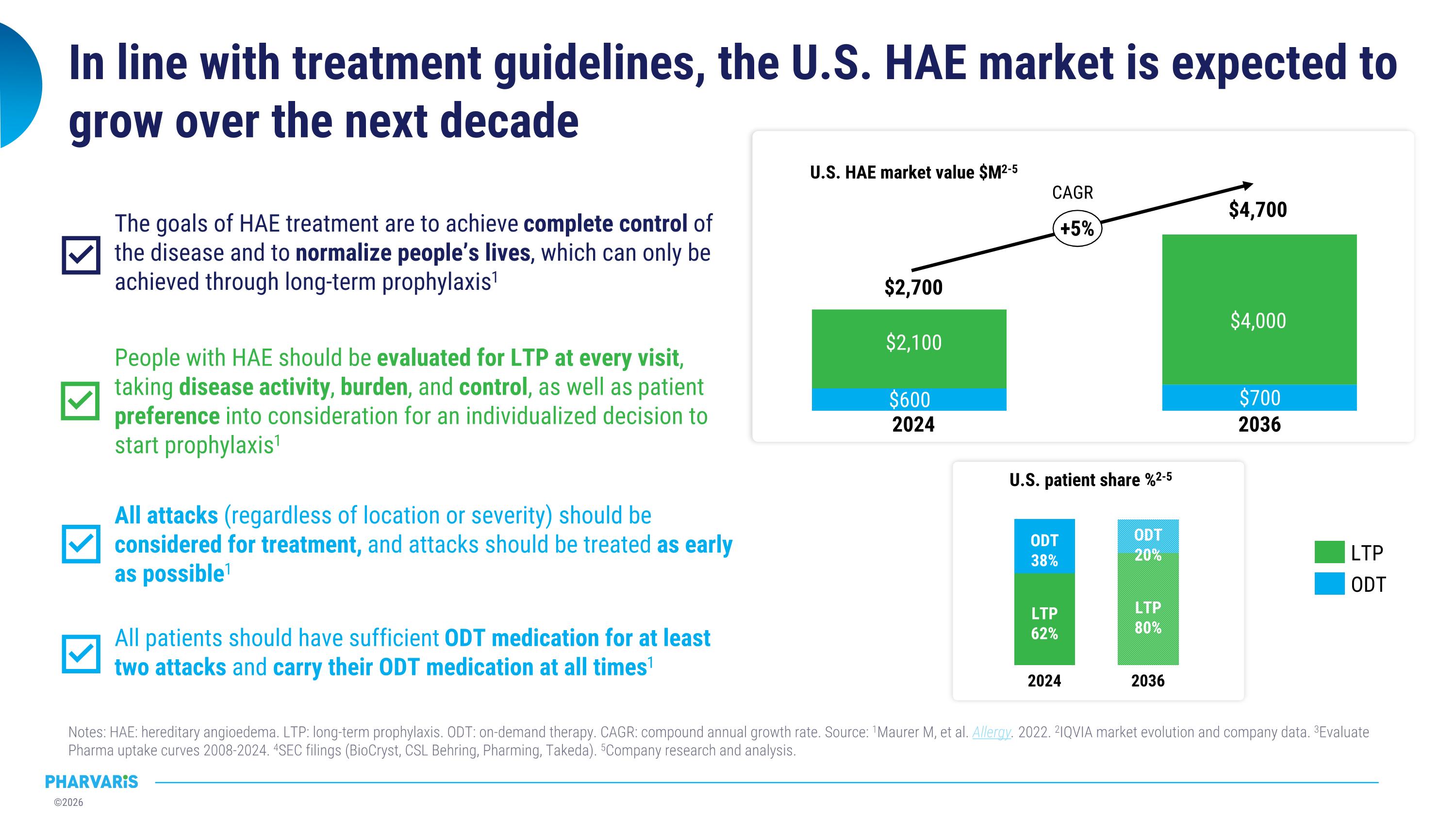
In line with treatment guidelines, the U.S. HAE market is expected to grow over the next decade Notes: HAE: hereditary angioedema. LTP: long-term prophylaxis. ODT: on-demand therapy. CAGR: compound annual growth rate. Source: 1Maurer M, et al. Allergy. 2022. 2IQVIA market evolution and company data. 3Evaluate Pharma uptake curves 2008-2024. 4SEC filings (BioCryst, CSL Behring, Pharming, Takeda). 5Company research and analysis. 2024 LTP 62% ODT 38% 2036 LTP 80% ODT 20% U.S. patient share %2-5 24% 2024 2036 $2,700 $4,700 $2,100 $4,000 +5% LTP ODT CAGR U.S. HAE market value $M2-5 All attacks (regardless of location or severity) should be considered for treatment, and attacks should be treated as early as possible1 All patients should have sufficient ODT medication for at least two attacks and carry their ODT medication at all times1 People with HAE should be evaluated for LTP at every visit, taking disease activity, burden, and control, as well as patient preference into consideration for an individualized decision to start prophylaxis1 The goals of HAE treatment are to achieve complete control of the disease and to normalize people’s lives, which can only be achieved through long-term prophylaxis1
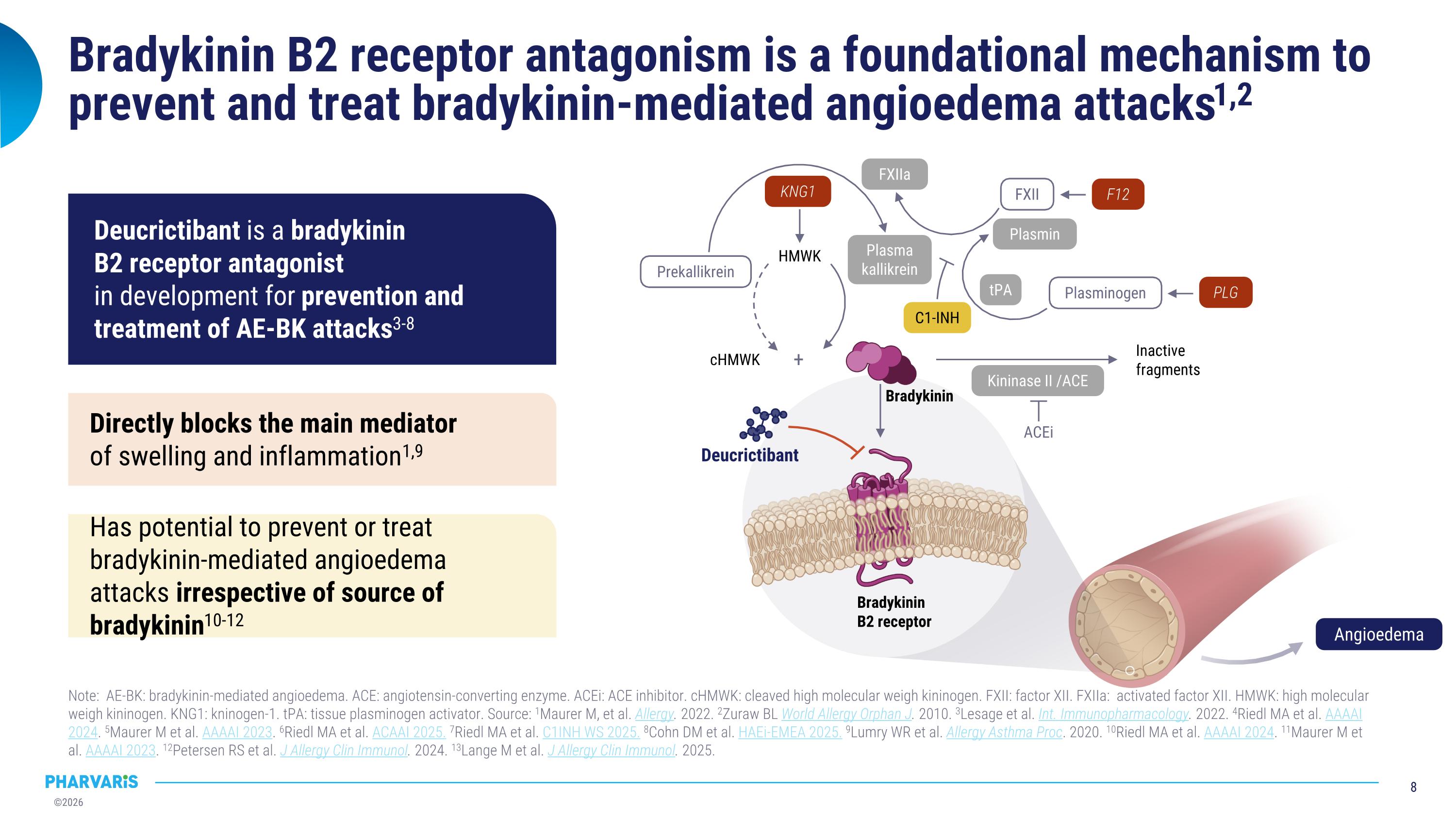
Bradykinin B2 receptor antagonism is a foundational mechanism to prevent and treat bradykinin-mediated angioedema attacks1,2 Note: AE-BK: bradykinin-mediated angioedema. ACE: angiotensin-converting enzyme. ACEi: ACE inhibitor. cHMWK: cleaved high molecular weigh kininogen. FXII: factor XII. FXIIa: activated factor XII. HMWK: high molecular weigh kininogen. KNG1: kninogen-1. tPA: tissue plasminogen activator. Source: 1Maurer M, et al. Allergy. 2022. 2Zuraw BL World Allergy Orphan J. 2010. 3Lesage et al. Int. Immunopharmacology. 2022. 4Riedl MA et al. AAAAI 2024. 5Maurer M et al. AAAAI 2023. 6Riedl MA et al. ACAAI 2025. 7Riedl MA et al. C1INH WS 2025. 8Cohn DM et al. HAEi-EMEA 2025. 9Lumry WR et al. Allergy Asthma Proc. 2020. 10Riedl MA et al. AAAAI 2024. 11Maurer M et al. AAAAI 2023. 12Petersen RS et al. J Allergy Clin Immunol. 2024. 13Lange M et al. J Allergy Clin Immunol. 2025. cHMWK Inactive fragments HMWK ACEi + Prekallikrein FXII Plasminogen Plasmin FXIIa tPA Kininase II /ACE Plasma kallikrein KNG1 F12 PLG C1-INH Bradykinin B2 receptor Deucrictibant Bradykinin Angioedema Has potential to prevent or treat bradykinin-mediated angioedema attacks irrespective of source of bradykinin10-12 Directly blocks the main mediator �of swelling and inflammation1,9 Deucrictibant is a bradykinin �B2 receptor antagonist �in development for prevention and treatment of AE-BK attacks3-8
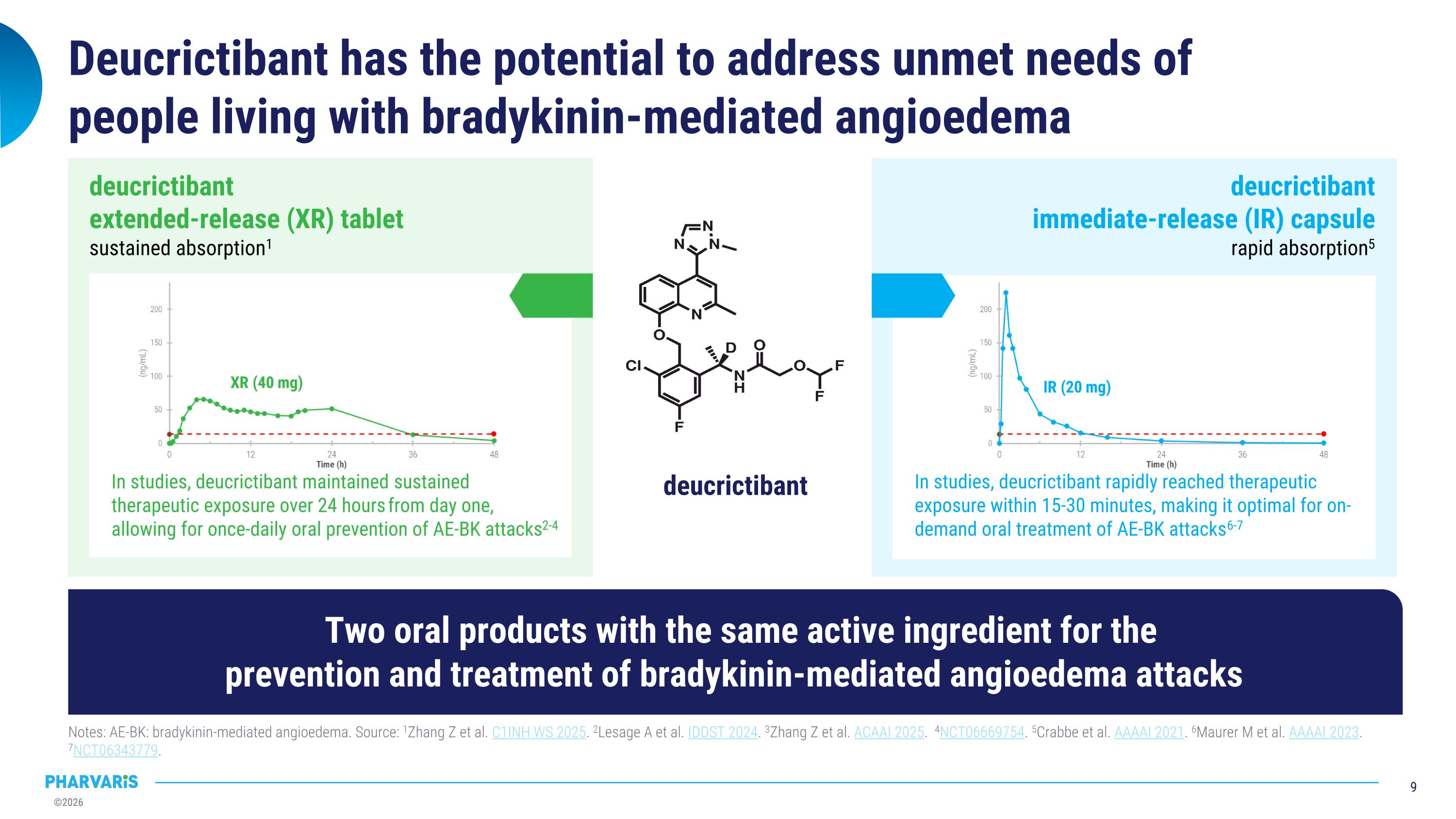
Deucrictibant has the potential to address unmet needs of�people living with bradykinin-mediated angioedema Notes: AE-BK: bradykinin-mediated angioedema. Source: 1Zhang Z et al. C1INH WS 2025. 2Lesage A et al. IDDST 2024. 3Zhang Z et al. ACAAI 2025. 4NCT06669754. 5Crabbe et al. AAAAI 2021. 6Maurer M et al. AAAAI 2023. 7NCT06343779. In studies, deucrictibant rapidly reached therapeutic exposure within 15-30 minutes, making it optimal for on-demand oral treatment of AE-BK attacks6-7 deucrictibant�immediate-release (IR) capsule�rapid absorption5 deucrictibant In studies, deucrictibant maintained sustained therapeutic exposure over 24 hours from day one, allowing for once-daily oral prevention of AE-BK attacks2-4 deucrictibant�extended-release (XR) tablet�sustained absorption1 Two oral products with the same active ingredient for the �prevention and treatment of bradykinin-mediated angioedema attacks

Deucrictibant differentiated profile for LTP and ODT ODT Oral LTP or ODT formulations Single oral pill Rapid to �steady state Rapid absorption Durable effective exposure Deucrictibant is the only HAE drug1 in development that allows for oral administration in both prophylaxis and on-demand2 Deucrictibant XR has the potential to achieve pharmacokinetic steady state within 2-3 days,5 providing protection against HAE attacks on the initial day3 of LTP initiation A durable effective exposure can potentially result in a high rate of �single-capsule attack resolution6,7 Deucrictibant IR rapidly reaches therapeutic exposure3 resulting in an end of progression™ in ~17.5 minutes,6 time to symptom relief in 1.28 hours,6 substantial symptom relief in 2.41 hours,6 and complete symptom relief in 11.28 hours6 Specific formulations with injectable-like efficacy™ allow for once-daily dosing3 (LTP) or rapid, single-capsule resolution3,4 of most HAE attacks (ODT) LTP Notes: AE-BK: bradykinin-mediated angioedema. LTP: long-term prophylaxis. ODT: on-demand therapy. XR: extended-release tablet formulation of deucrictibant. IR: immediate-release capsule formulation of deucrictibant. The terms injectable-like efficacy and End of Progression are registered trademarks of Pharvaris GmbH.�Sources: 1Company research. 2Lesage et al. IDDST 2024. 3Zhang Z et al. ACAAI 2025 4Anderson J et al. ACAAI 2025. 5Maurer M et al. HAEi Workshop 2022. 6RAPIDe-3 topline data. All data reported are median times. 7Riedl MA et al. C1INH WS 2025.
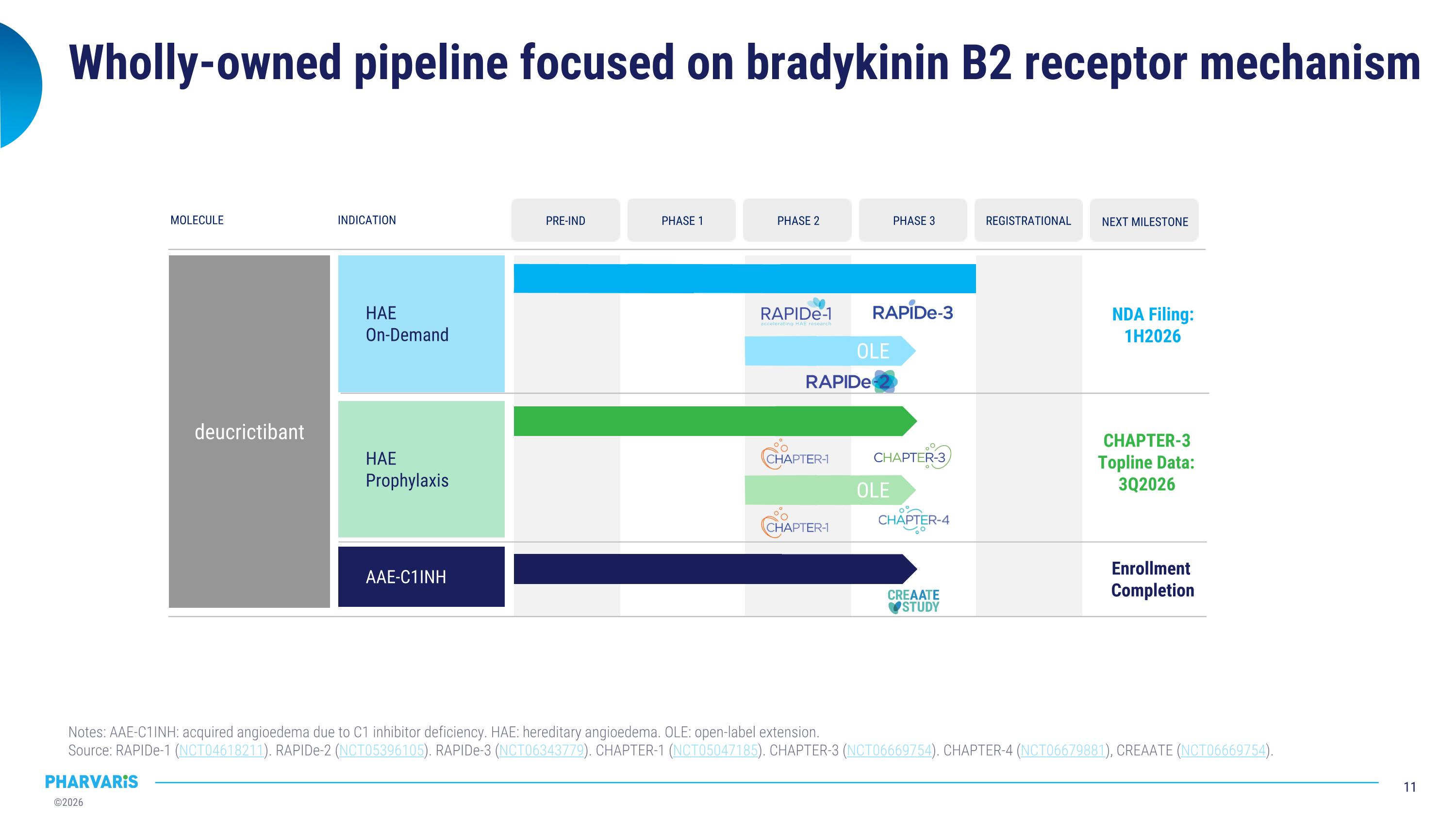
Wholly-owned pipeline focused on bradykinin B2 receptor mechanism Notes: AAE-C1INH: acquired angioedema due to C1 inhibitor deficiency. HAE: hereditary angioedema. OLE: open-label extension. �Source: RAPIDe-1 (NCT04618211). RAPIDe-2 (NCT05396105). RAPIDe-3 (NCT06343779). CHAPTER-1 (NCT05047185). CHAPTER-3 (NCT06669754). CHAPTER-4 (NCT06679881), CREAATE (NCT06669754). MOLECULE INDICATION deucrictibant HAE On-Demand HAE Prophylaxis NDA Filing: 1H2026 AAE-C1INH PRE-IND PHASE 1 PHASE 2 PHASE 3 REGISTRATIONAL NEXT MILESTONE OLE CHAPTER-3�Topline Data: �3Q2026 OLE Enrollment �Completion

HAE On-Demand Treatment (ODT) HAE Long-Term Prophylaxis (LTP) Deucrictibant clinical development program Notes: HAE: hereditary angioedema. OLE: open-label extension. AAE-C1INH: acquired angioedema due to C1 inhibitor deficiency. Source: 1NCT05047185. 2NCT06669754. 3NCT06679881. 4NCT04618211. 5NCT05396105. 6NCT06343779. 7NCT06669754. HAE Phase 24 Complete HAE Phase 2/3 OLE5 Ongoing HAE Phase 3 pivotal6 Complete HAE Phase 21 Complete HAE Phase 3 pivotal2 Ongoing HAE Phase 3 OLE3 Ongoing AAE-C1INH7 Part 1: AAE-C1INH LTP pivotal Part 2: AAE-C1INH ODT pivotal Part 3: AAE-C1INH ODT OLE Ongoing
On-Demand Deucrictibant immediate-release capsules

People with HAE want effective on-demand therapies in a �well-tolerated, convenient oral form Notes: HAE: Hereditary Angioedema. ODT: On-Demand Treatment. Sources: 1Betschel SD et al. Allergy Asthma Clin Immunol. 2024. 2Christiansen S et al. Ann Allergy Asthma Immunol. 2024. 3Bernstein JA et al. J Manag Care Spec Pharm. 2020. 4Mendivil J et al., Allergy Asthma Clin Immunol. 2023. 5Data on File. …and are less cumbersome to administer… …so there are no reasons to delay treatment for attacks “severe enough” to treat redosing rates with currently available ODT3 Up to 44% Patients want an option that is highly effective… 32% saved doses for severe attacks2 Up to 28% report injections are too painful4 of patients delay treating their HAE attacks1, which lead to attacks that… Up to 86% are ~2x more likely to increase in severity over time (with >one-hour delay)2 last up to 4x longer (with >eight-hour delay)2 50% Up to 50% of patients do NOT carry rescue medication with them5
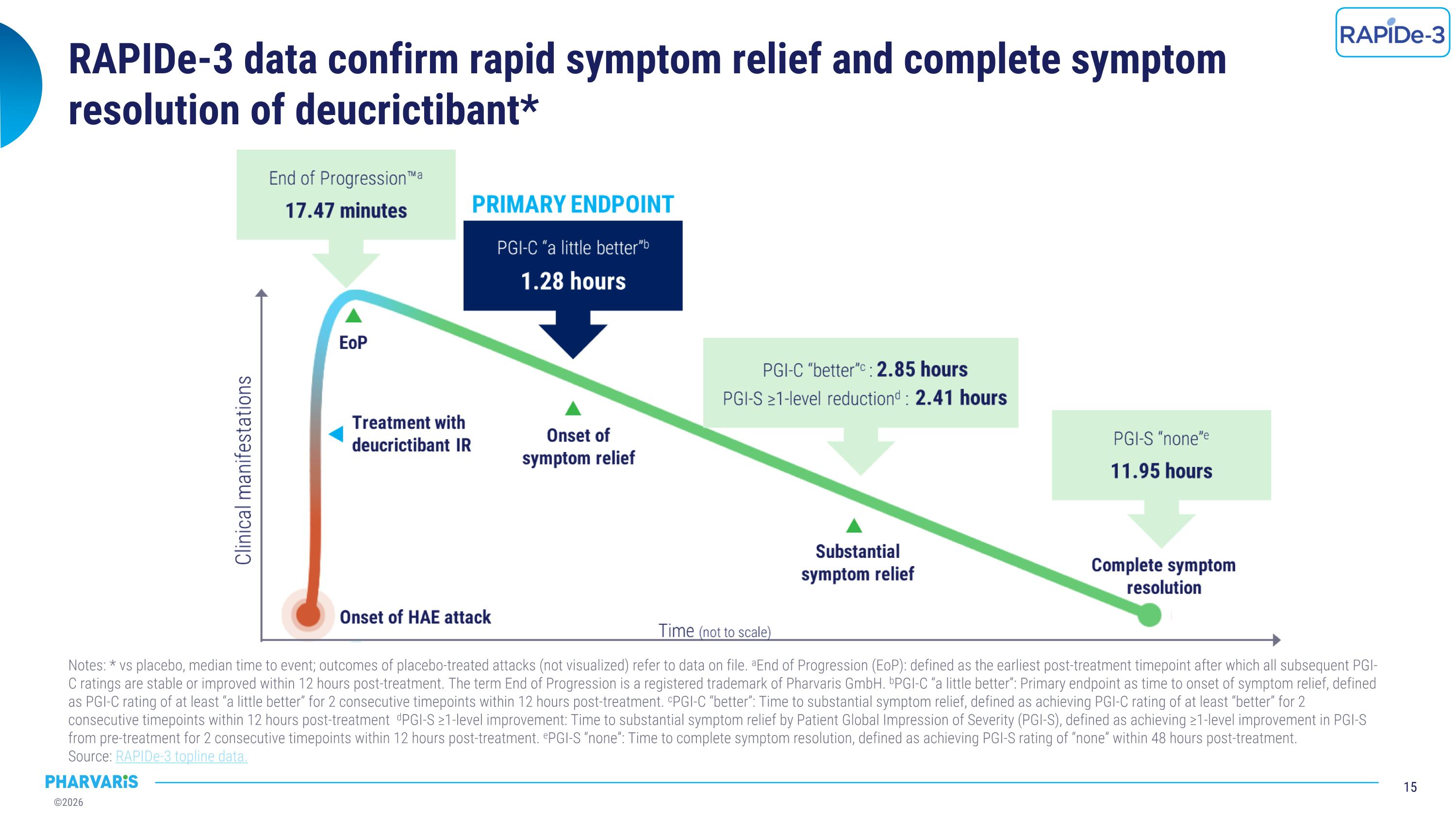
RAPIDe-3 data confirm rapid symptom relief and complete symptom resolution of deucrictibant* Notes: * vs placebo, median time to event; outcomes of placebo-treated attacks (not visualized) refer to data on file. aEnd of Progression (EoP): defined as the earliest post-treatment timepoint after which all subsequent PGI-C ratings are stable or improved within 12 hours post-treatment. The term End of Progression is a registered trademark of Pharvaris GmbH. bPGI-C “a little better”: Primary endpoint as time to onset of symptom relief, defined as PGI-C rating of at least “a little better” for 2 consecutive timepoints within 12 hours post-treatment. cPGI-C “better”: Time to substantial symptom relief, defined as achieving PGI-C rating of at least “better” for 2 consecutive timepoints within 12 hours post-treatment dPGI-S ≥1-level improvement: Time to substantial symptom relief by Patient Global Impression of Severity (PGI-S), defined as achieving ≥1-level improvement in PGI-S from pre-treatment for 2 consecutive timepoints within 12 hours post-treatment. ePGI-S “none”: Time to complete symptom resolution, defined as achieving PGI-S rating of “none” within 48 hours post-treatment. �Source: RAPIDe-3 topline data.
Long-Term Prophylaxis Deucrictibant extended-release tablets
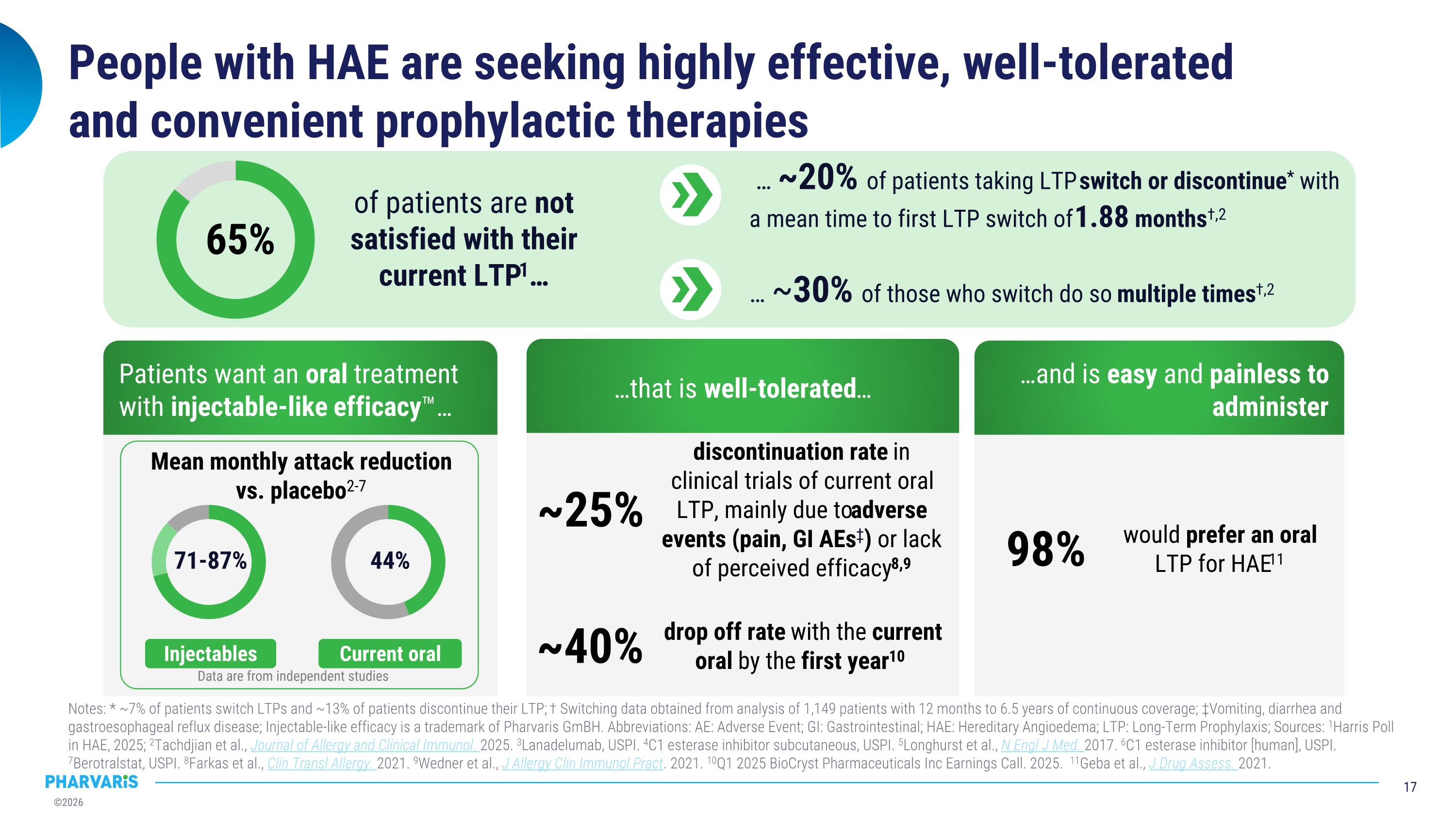
People with HAE are seeking highly effective, well-tolerated�and convenient prophylactic therapies Notes: * ~7% of patients switch LTPs and ~13% of patients discontinue their LTP; † Switching data obtained from analysis of 1,149 patients with 12 months to 6.5 years of continuous coverage; ‡Vomiting, diarrhea and gastroesophageal reflux disease; Injectable-like efficacy is a trademark of Pharvaris GmBH. Abbreviations: AE: Adverse Event; GI: Gastrointestinal; HAE: Hereditary Angioedema; LTP: Long-Term Prophylaxis; Sources: 1Harris Poll in HAE, 2025; 2Tachdjian et al., Journal of Allergy and Clinical Immunol. 2025. 3Lanadelumab, USPI. 4C1 esterase inhibitor subcutaneous, USPI. 5Longhurst et al., N Engl J Med. 2017. 6C1 esterase inhibitor [human], USPI. 7Berotralstat, USPI. 8Farkas et al., Clin Transl Allergy. 2021. 9Wedner et al., J Allergy Clin Immunol Pract. 2021. 10Q1 2025 BioCryst Pharmaceuticals Inc Earnings Call. 2025. 11Geba et al., J Drug Assess. 2021. Current oral Injectables Mean monthly attack reduction vs. placebo2-7 71-87% 44% discontinuation rate in clinical trials of current oral LTP, mainly due to adverse events (pain, GI AEs‡) or lack of perceived efficacy8,9 ~25% would prefer an oral LTP for HAE11 98% Patients want an oral treatment with injectable-like efficacy™… …that is well-tolerated… …and is easy and painless to administer 65% … ~20% of patients taking LTP switch or discontinue* with a mean time to first LTP switch of 1.88 months†,2 of patients are not satisfied with their current LTP1… … ~30% of those who switch do so multiple times†,2 drop off rate with the current oral by the first year10 ~40% Data are from independent studies
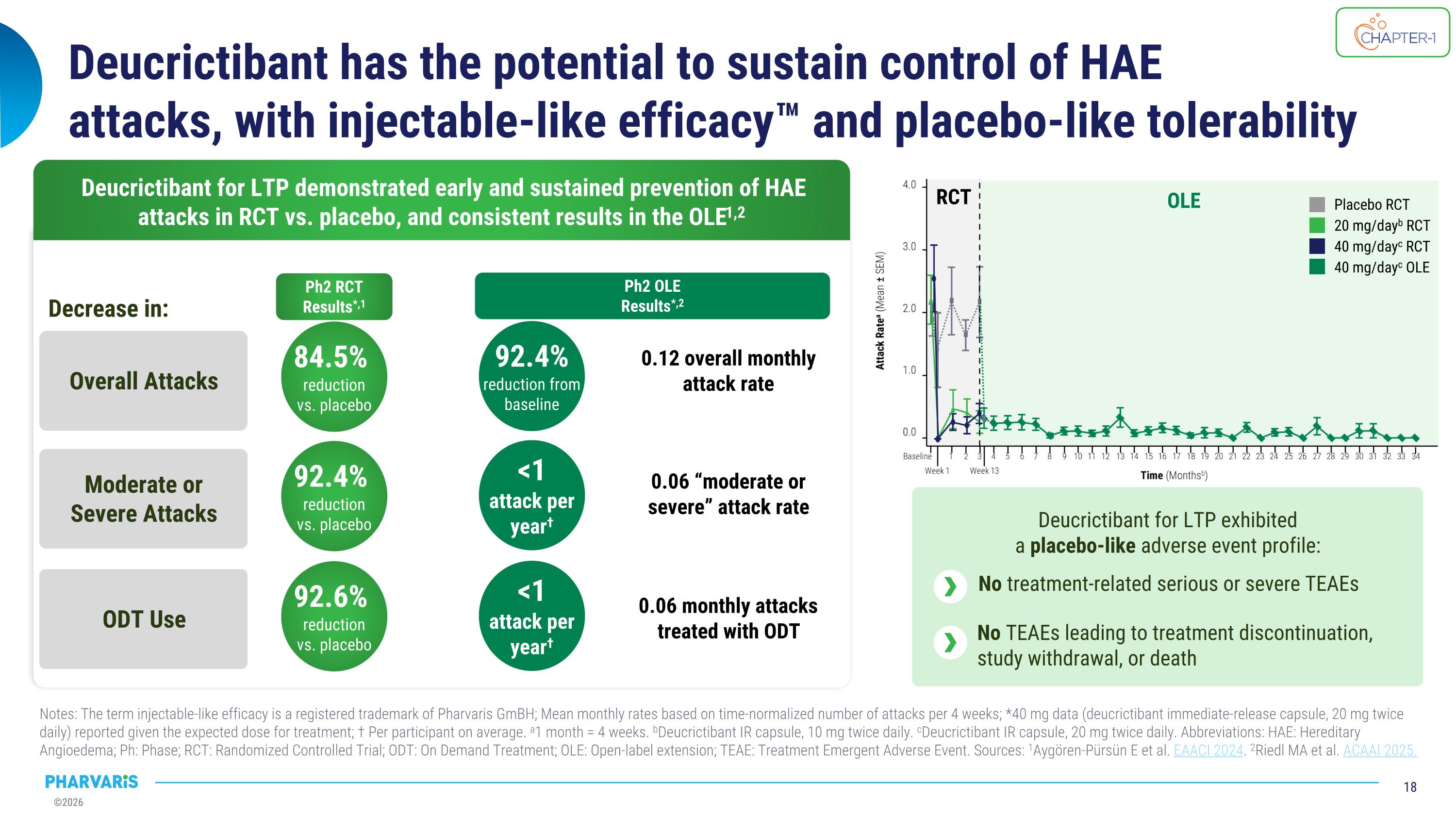
Deucrictibant has the potential to sustain control of HAE �attacks, with injectable-like efficacy™ and placebo-like tolerability Notes: The term injectable-like efficacy is a registered trademark of Pharvaris GmBH; Mean monthly rates based on time-normalized number of attacks per 4 weeks; *40 mg data (deucrictibant immediate-release capsule, 20 mg twice daily) reported given the expected dose for treatment; † Per participant on average. a1 month = 4 weeks. bDeucrictibant IR capsule, 10 mg twice daily. cDeucrictibant IR capsule, 20 mg twice daily. Abbreviations: HAE: Hereditary Angioedema; Ph: Phase; RCT: Randomized Controlled Trial; ODT: On Demand Treatment; OLE: Open-label extension; TEAE: Treatment Emergent Adverse Event. Sources: 1Aygören-Pürsün E et al. EAACI 2024. 2Riedl MA et al. ACAAI 2025. Deucrictibant for LTP exhibited�a placebo-like adverse event profile: No treatment-related serious or severe TEAEs No TEAEs leading to treatment discontinuation, study withdrawal, or death Deucrictibant for LTP demonstrated early and sustained prevention of HAE attacks in RCT vs. placebo, and consistent results in the OLE1,2 Overall Attacks Moderate or Severe Attacks ODT Use Ph2 RCT Results*,1 Ph2 OLE Results*,2 <1 attack per year† <1 attack per year† 84.5% reduction vs. placebo 92.4% reduction from baseline 92.4% reduction vs. placebo 92.6% reduction vs. placebo 0.12 overall monthly attack rate 0.06 “moderate or severe” attack rate 0.06 monthly attacks treated with ODT Decrease in: RCT OLE 40 mg/dayc OLE 40 mg/dayc RCT 20 mg/dayb RCT Placebo RCT
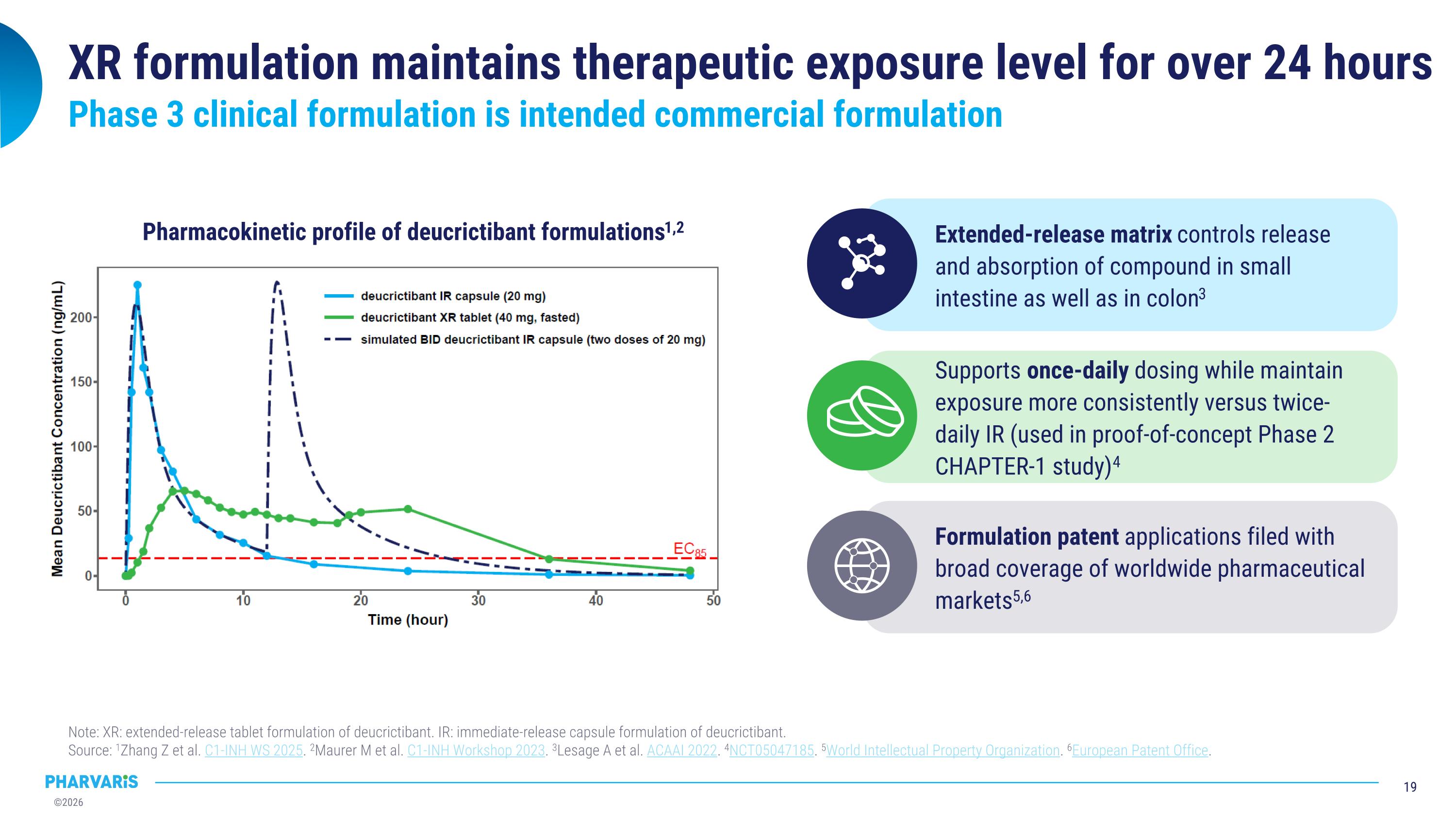
XR formulation maintains therapeutic exposure level for over 24 hours Note: XR: extended-release tablet formulation of deucrictibant. IR: immediate-release capsule formulation of deucrictibant. �Source: 1Zhang Z et al. C1-INH WS 2025. 2Maurer M et al. C1-INH Workshop 2023. 3Lesage A et al. ACAAI 2022. 4NCT05047185. 5World Intellectual Property Organization. 6European Patent Office. Pharmacokinetic profile of deucrictibant formulations1,2 Extended-release matrix controls release and absorption of compound in small intestine as well as in colon3 Formulation patent applications filed with broad coverage of worldwide pharmaceutical markets5,6 Supports once-daily dosing while maintain exposure more consistently versus twice-daily IR (used in proof-of-concept Phase 2 CHAPTER-1 study)4 Phase 3 clinical formulation is intended commercial formulation
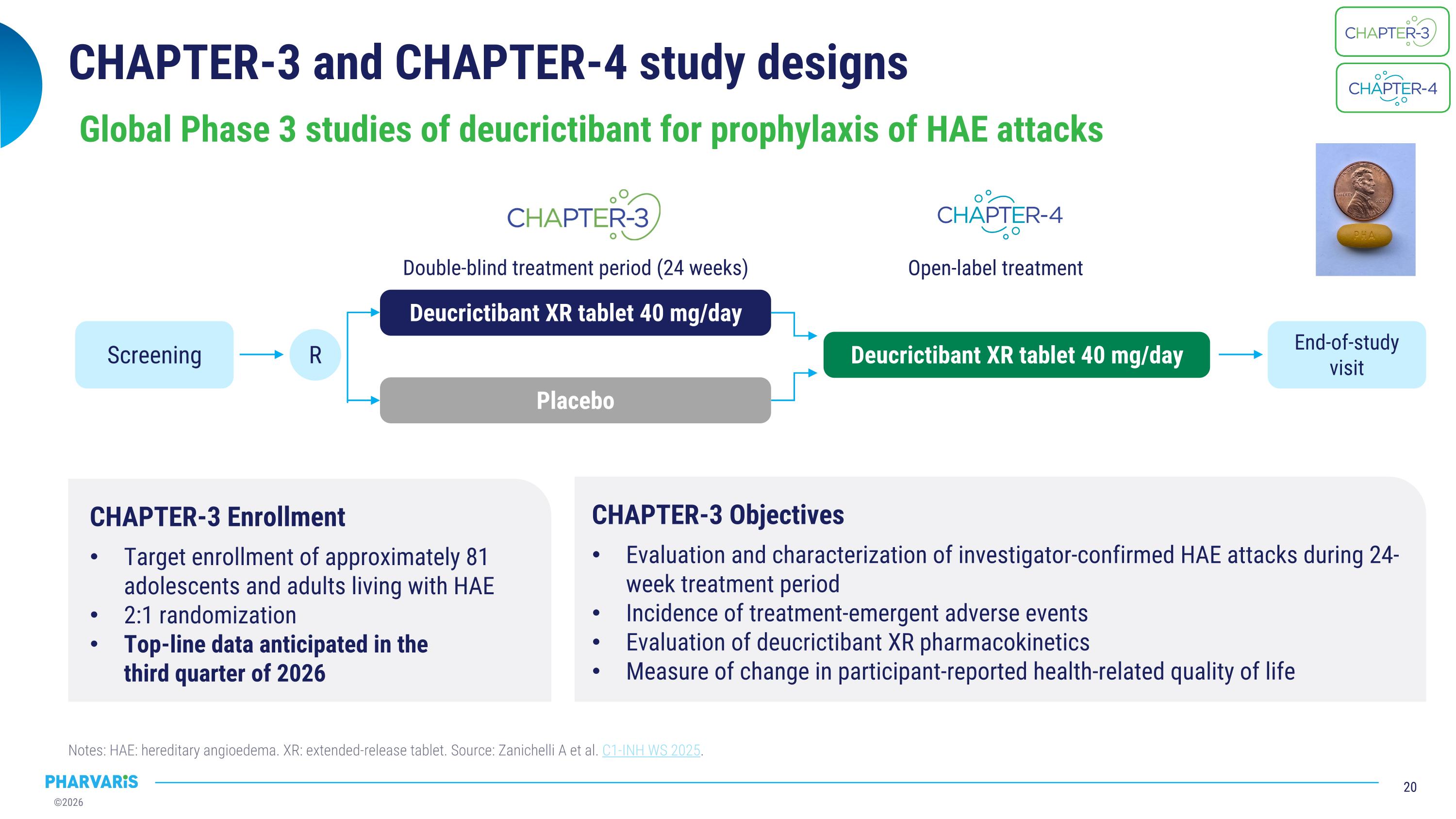
CHAPTER-3 and CHAPTER-4 study designs Notes: HAE: hereditary angioedema. XR: extended-release tablet. Source: Zanichelli A et al. C1-INH WS 2025. Global Phase 3 studies of deucrictibant for prophylaxis of HAE attacks CHAPTER-3 Enrollment Target enrollment of approximately 81 adolescents and adults living with HAE 2:1 randomization Top-line data anticipated in the �third quarter of 2026 CHAPTER-3 Objectives Evaluation and characterization of investigator-confirmed HAE attacks during 24-week treatment period Incidence of treatment-emergent adverse events Evaluation of deucrictibant XR pharmacokinetics Measure of change in participant-reported health-related quality of life R Deucrictibant XR tablet 40 mg/day Deucrictibant XR tablet 40 mg/day End-of-study visit Screening Double-blind treatment period (24 weeks) Open-label treatment Placebo

Acquired Angioedema due to �C1-Inhibitor Deficiency �(AAE-C1INH)
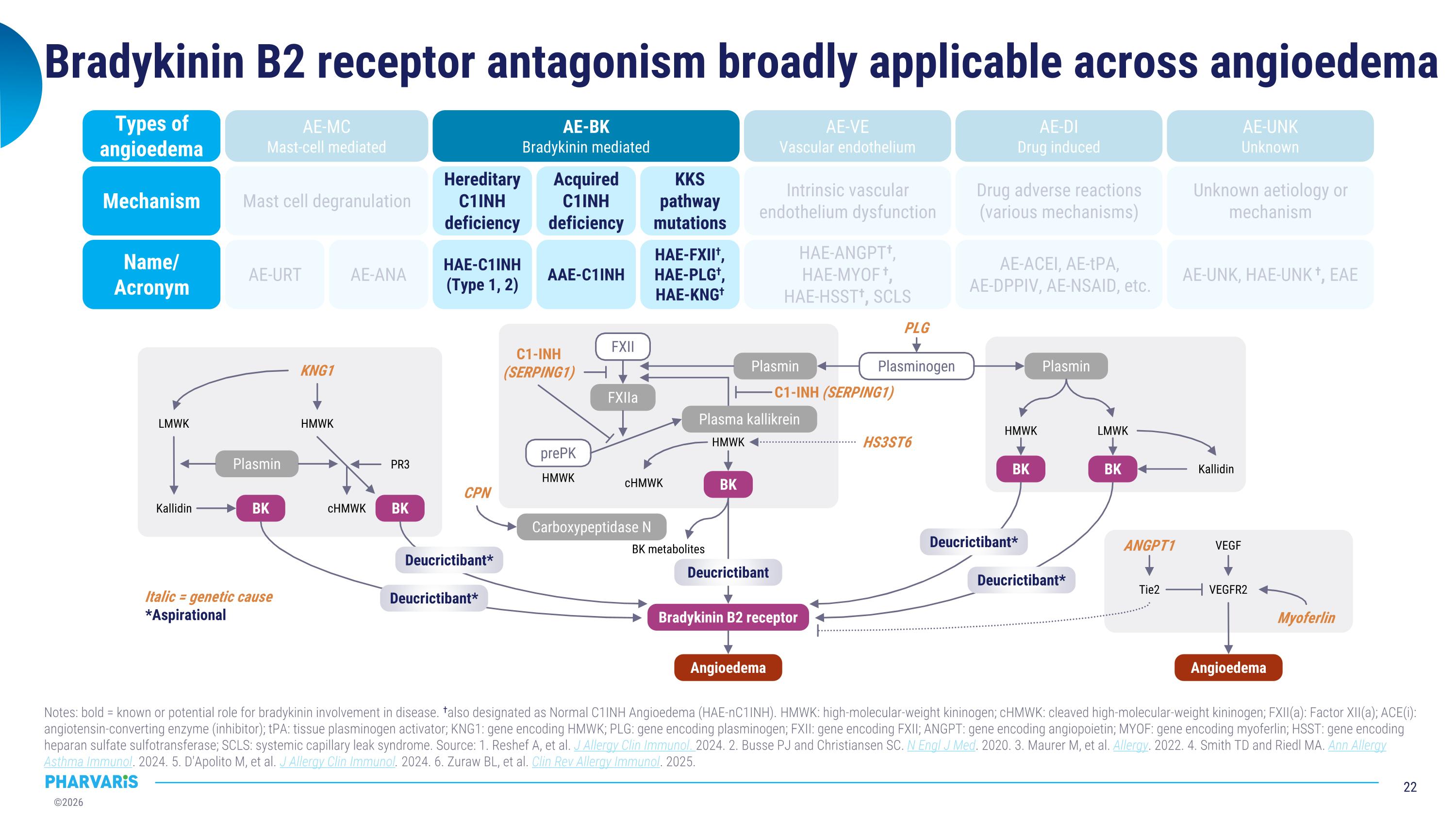
Bradykinin B2 receptor antagonism broadly applicable across angioedema Notes: bold = known or potential role for bradykinin involvement in disease. †also designated as Normal C1INH Angioedema (HAE-nC1INH). HMWK: high-molecular-weight kininogen; cHMWK: cleaved high-molecular-weight kininogen; FXII(a): Factor XII(a); ACE(i): angiotensin-converting enzyme (inhibitor); tPA: tissue plasminogen activator; KNG1: gene encoding HMWK; PLG: gene encoding plasminogen; FXII: gene encoding FXII; ANGPT: gene encoding angiopoietin; MYOF: gene encoding myoferlin; HSST: gene encoding heparan sulfate sulfotransferase; SCLS: systemic capillary leak syndrome. Source: 1. Reshef A, et al. J Allergy Clin Immunol. 2024. 2. Busse PJ and Christiansen SC. N Engl J Med. 2020. 3. Maurer M, et al. Allergy. 2022. 4. Smith TD and Riedl MA. Ann Allergy Asthma Immunol. 2024. 5. D'Apolito M, et al. J Allergy Clin Immunol. 2024. 6. Zuraw BL, et al. Clin Rev Allergy Immunol. 2025. AE-URT AE-ANA HAE-C1INH (Type 1, 2) AAE-C1INH HAE-FXII†, HAE-PLG†, HAE-KNG† HAE-ANGPT†, �HAE-MYOF †, �HAE-HSST†, SCLS AE-ACEI, AE-tPA, �AE-DPPIV, AE-NSAID, etc. AE-UNK, HAE-UNK †, EAE Mast cell degranulation Hereditary C1INH deficiency Acquired C1INH deficiency KKS pathway mutations Intrinsic vascular endothelium dysfunction Drug adverse reactions (various mechanisms) Unknown aetiology or mechanism AE-MC Mast-cell mediated AE-BK Bradykinin mediated AE-VE Vascular endothelium AE-DI Drug induced AE-UNK Unknown Name/�Acronym Mechanism Types of�angioedema Italic = genetic cause *Aspirational Angioedema Bradykinin B2 receptor Angioedema BK BK BK BK BK Deucrictibant Deucrictibant* Deucrictibant* Deucrictibant* Deucrictibant* Plasmin Plasmin Plasmin FXIIa Carboxypeptidase N BK metabolites FXII Plasminogen prePK HMWK cHMWK HMWK C1-INH (SERPING1) C1-INH�(SERPING1) HS3ST6 PLG Plasma kallikrein HMWK LMWK Kallidin VEGFR2 VEGF Tie2 Myoferlin ANGPT1 CPN KNG1 HMWK LMWK Kallidin PR3 cHMWK
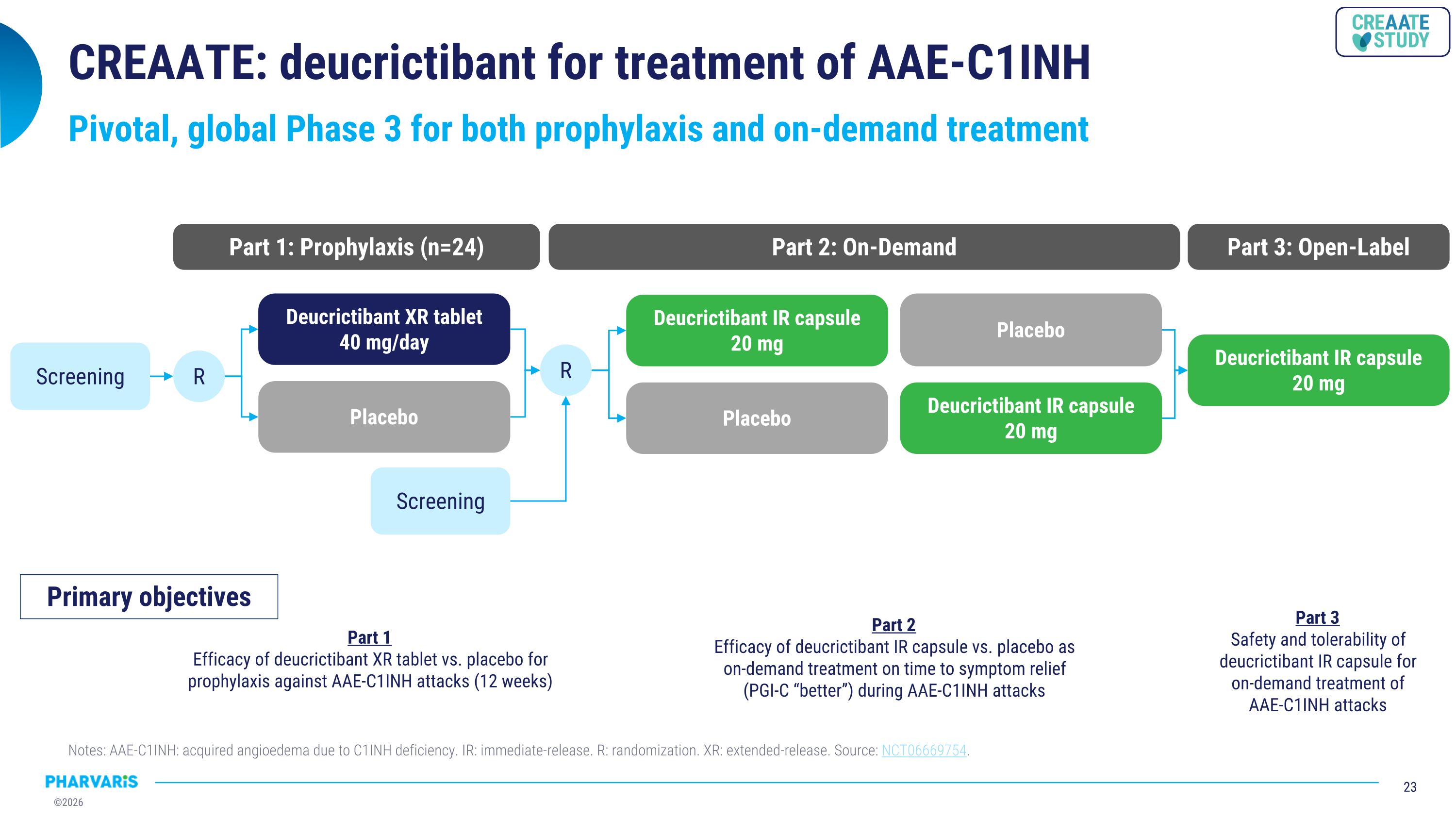
CREAATE: deucrictibant for treatment of AAE-C1INH Pivotal, global Phase 3 for both prophylaxis and on-demand treatment R Deucrictibant XR tablet 40 mg/day Screening Placebo R Deucrictibant IR capsule 20 mg Placebo Deucrictibant IR capsule 20 mg Placebo Deucrictibant IR capsule 20 mg Screening Part 1: Prophylaxis (n=24) Part 2: On-Demand Part 3: Open-Label Primary objectives Part 2 Efficacy of deucrictibant IR capsule vs. placebo as on-demand treatment on time to symptom relief �(PGI-C “better”) during AAE-C1INH attacks Part 1 Efficacy of deucrictibant XR tablet vs. placebo for prophylaxis against AAE-C1INH attacks (12 weeks) Part 3 Safety and tolerability of deucrictibant IR capsule for on-demand treatment of AAE-C1INH attacks Notes: AAE-C1INH: acquired angioedema due to C1INH deficiency. IR: immediate-release. R: randomization. XR: extended-release. Source: NCT06669754.

Pharvaris Vision
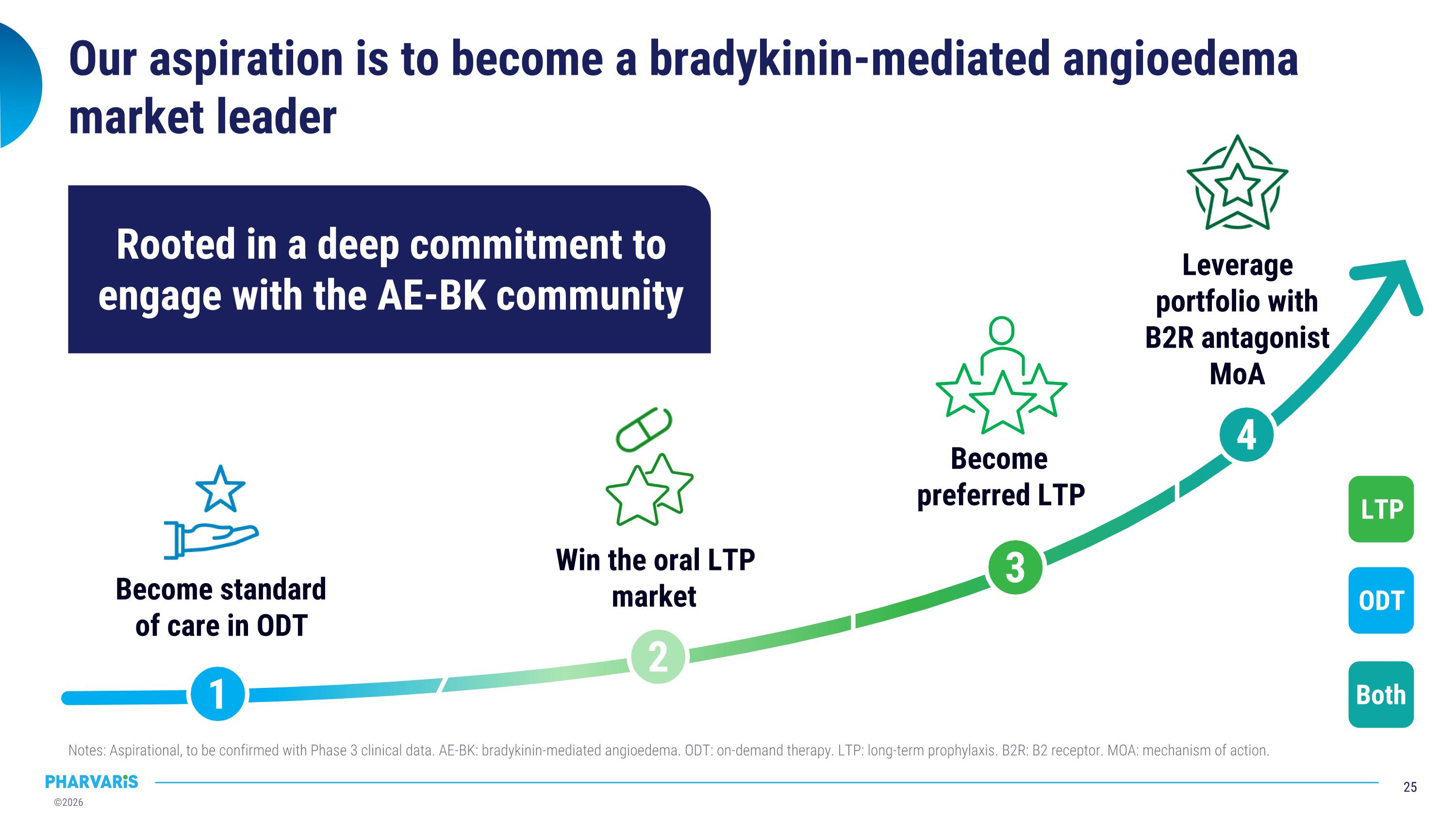
Our aspiration is to become a bradykinin-mediated angioedema market leader Notes: Aspirational, to be confirmed with Phase 3 clinical data. AE-BK: bradykinin-mediated angioedema. ODT: on-demand therapy. LTP: long-term prophylaxis. B2R: B2 receptor. MOA: mechanism of action. 1 2 4 3 Win the oral LTP market Become preferred LTP Leverage �portfolio with �B2R antagonist MoA Become standard of care in ODT LTP ODT Both Rooted in a deep commitment to engage with the AE-BK community

www.pharvaris.com NASDAQ: PHVS



























